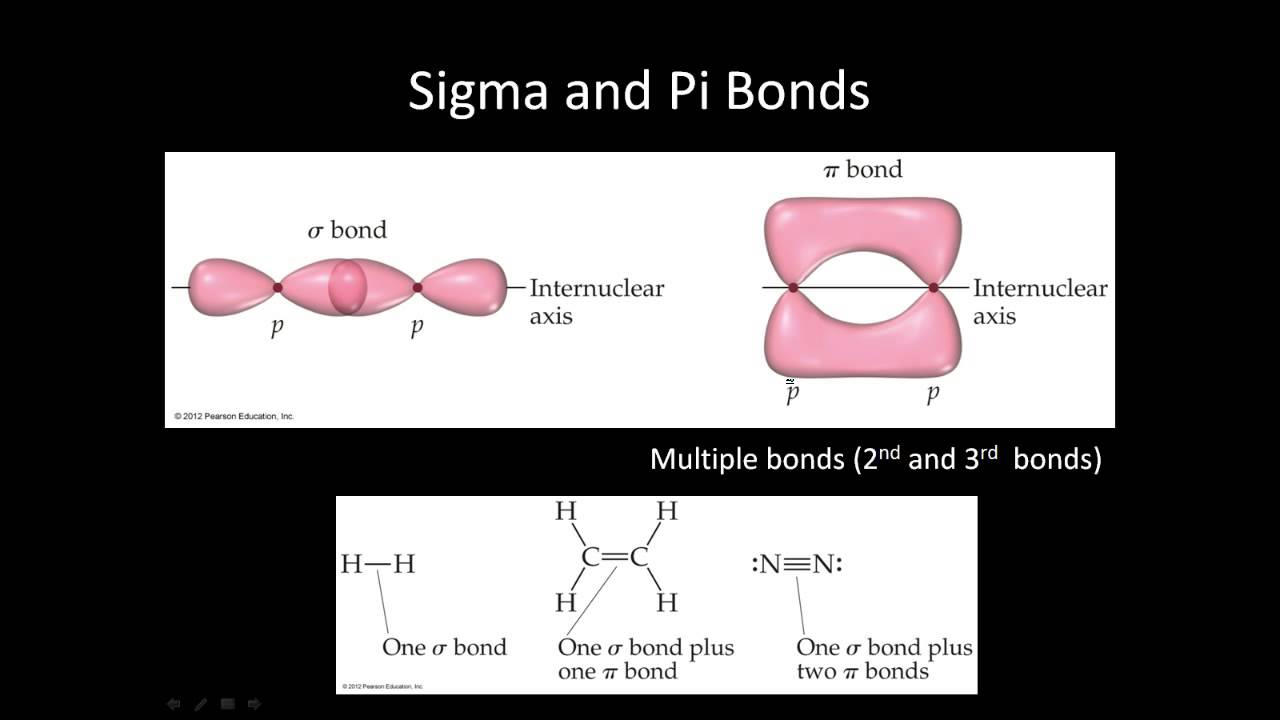
In any bond, the first two electrons create a sigma bond. A pi bond is created when two more electrons share the space around a sigma bond. 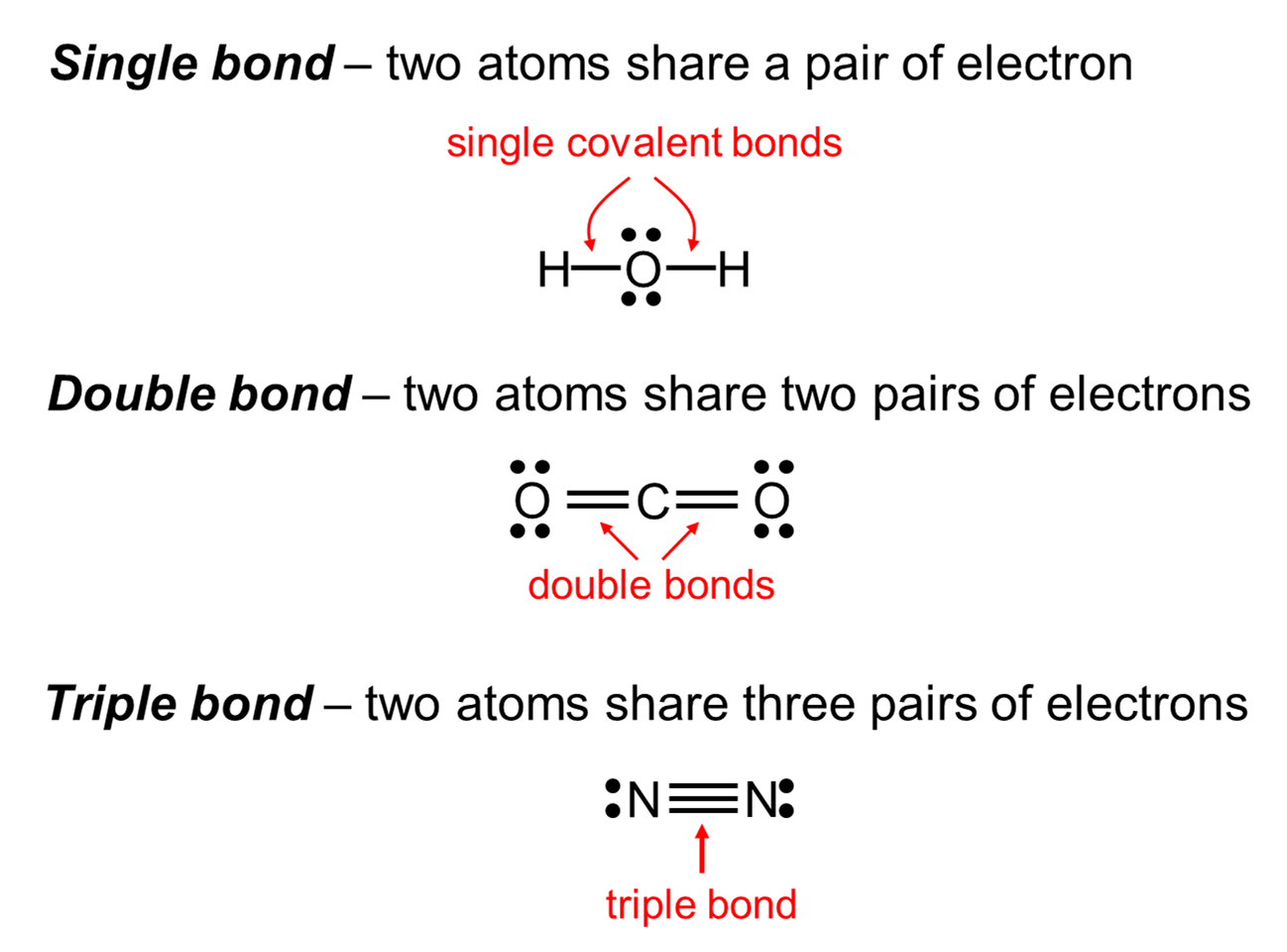
The principles involved - promotion of electrons if necessary, then hybridisation, followed by the formation of molecular orbitals - can be applied to any covalently-bound molecule. A sigma bond is created when two electrons share the space between two atoms in a bond. Each orbital holds the 2 electrons that we've previously drawn as a dot and a cross. When a covalent bond is formed, the atomic orbitals (the orbitals in the individual atoms) merge to produce a new molecular orbital which contains the electron pair which creates the bond.įour molecular orbitals are formed, looking rather like the original sp 3 hybrids, but with a hydrogen nucleus embedded in each lobe. Remember that hydrogen's electron is in a 1s orbital - a spherically symmetric region of space surrounding the nucleus where there is some fixed chance (say 95%) of finding the electron. For clarity, the nucleus is drawn far larger than it really is. You can picture the nucleus as being at the centre of a tetrahedron (a triangularly based pyramid) with the orbitals pointing to the corners. Sp 3 hybrid orbitals look a bit like half a p orbital, and they arrange themselves in space so that they are as far apart as possible. In how many sigma bonds does the highlighted atom participate b. In this case the unhybridized p orbital is of not much relevance. A sigma bond is a type of covalent chemical bond that results from the formation of a molecular orbital by the end to end overlap of the atomic orbitals. You should read "sp 3" as "s p three" - not as "s p cubed". ( 83 votes) Flag Ganesh D 8 years ago Basically an orbital (be it s,p,d or f) is merely an area where there is a probability of finding an electron. This reorganizes the electrons into four identical hybrid orbitals called sp 3 hybrids (because they are made from one s orbital and three p orbitals). Sigma Bonds (4 images) (Drag and drop into the appropriate area) Increasing Bond Length. The electrons rearrange themselves again in a process called hybridization. Drag the bonds (A, B, C, and D) into order from shortest length to longest.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
